LUYOR-3109高强度紫外催化光源促销
LUYOR-3109紫外光源采用了9颗365nm大功率led,安装有二次光学透镜,输出紫外线强度高,...
2024-08-08作者:生命科学事业部时间:2019-10-07 15:50:49浏览17839 次
红色荧光蛋白(RFP)是从太平洋与海葵相关的 (Discosomasp)中分离出来的一种能在紫外线的照射下可发射红色荧光的蛋白。在细胞中荧光转换效率高.是目前发现的具有最长激发波长与发射波长的自然发光荧光蛋白,RFP基因编码的蛋白质由225个氨基酸组成,相对分子质量(Mr)为25.9ku.RFP以被广泛用于动物、植物和酵母等真核细胞内基因表达,其基因在原核细胞中的应用也越来越受关注
红色荧光蛋白(RFP)是从太平洋与海葵相关的 (Discosomasp)中分离出来的一种能在紫外线的照射下可发射红色荧光的蛋白。在细胞中荧光转换效率高.是目前发现的具有最长激发波长与发射波长的自然发光荧光蛋白,RFP基因编码的蛋白质由225个氨基酸组成,相对分子质量(Mr)为25.9ku.RFP以被广泛用于动物、植物和酵母等真核细胞内基因表达,其基因在原核细胞中的应用也越来越受关注。红色荧光蛋白RFP是的一种生物发光蛋白,由DsRed和增强型黄色荧光蛋白(CFP),所以RFP作为一种新型可视性标签在真核基因表达、目的蛋白的细胞及亚细胞定位等研究中得到广泛应用 。然而,RFP的缺点是需要形成结构稳定的同源四聚体、且需要紫外光等特殊光源的激发才能发射出红色荧光,尤其是RFP容易形成难溶性多聚体 。DsRed2是源于野生型DsRed的一种人工突变基因,该突变基因的表达产物RFP2,其蛋白结构与绿色荧光蛋白(GFP)相似;与DSRED基因编码的RFP 蛋白(氨基酸残基数为248)相比,RFP2蛋白氨基酸序列的C⁃末端少23 个氨基酸,其发光基团的成熟速度更快,形成的难溶性多聚合体也有所减少。
红色荧光蛋白激发光与发射光波长:
如何选择合适的RFP(DsRed)标签抗体?
我们在选择RFP(DsRed)抗体时,主要根据自己的需要,特别是实验应用上的需要。另外,一个抗体检测后的应用类型越多,在使用过程中的选择余地就越大;而小鼠源的单克隆抗体一般在特异性、稳定性以及效价都要优于多克隆抗体。 另外,抗体的效价也是考察该抗体性价比的重要方面,也即相当于实际应用时的稀释比率。一个效价高的100μl RFP抗体(假如其WB检测的稀释比率是1:5000,最终使用液相当于500ml),要比效价低的1ml的RFP抗体(假如WB检测的稀释比率是 1:200,最终使用液相当于200ml)性价比更高。
如果要观察Dsred红色荧光蛋白(red Fluorescent Protein)的表达,美国路阳生产的便携式荧光蛋白激发光源可以选择LUYOR-3260GR和LUYOR-3415(X)G系列双波长荧光蛋白激发光源。红色荧光蛋白采用绿光激发,佩戴LUV-50A红色观察眼镜观察,如希望提供更多详细信息,可直接联系上海路阳生物技术有限公司的销售客服。

上图为LUYOR-3415RG便携式红色荧光蛋白激发光源

上图为LUV-50A红色荧光蛋白观察眼镜

上图为LUV-590A 红色荧光蛋白拍照滤镜
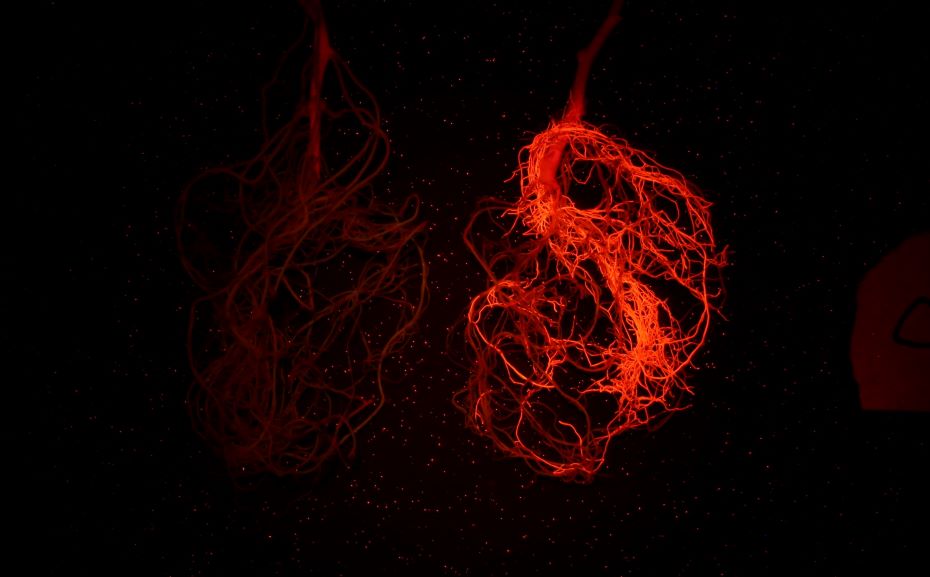
上图为红色荧光蛋白在大豆根系上的表达(LUYOR-3415RG照射,LUV-590A滤镜拍摄)
A major goal of fluorescent protein development has become the construction of a red-emitting derivative that equals or exceeds the advanced properties of enhanced green fluorescent protein. Among the advantages of a suitable red fluorescent protein are the potential compatibility with existing confocal and widefield microscopes (and their filter sets), along with an increased capacity to image entire animals, which are significantly more transparent to red light. Because the construction of red-shifted mutants from the Aequorea victoria jellyfish green fluorescent protein beyond the yellow spectral region has proven largely unsuccessful, investigators have turned their search to the tropical reef corals.
The first coral-derived fluorescent protein to be extensively utilized was derived from Discosoma striata and is commonly referred to as DsRed. Once fully matured, the fluorescence emission spectrum of DsRed features a peak at 583 nanometers whereas the excitation spectrum has a major peak at 558 nanometers and a minor peak around 500 nanometers. Several problems are associated with using DsRed, however. Maturation of DsRed fluorescence occurs slowly and proceeds through a time period when fluorescence emission is in the green region. Termed the green state, this artifact has proven problematic for multiple labeling experiments with other green fluorescent proteins because of the spectral overlap. Furthermore, DsRed is an obligate tetramer and can form large protein aggregates in living cells. Although these features are inconsequential for the use of DsRed as a reporter of gene expression, the usefulness of DsRed as an epitope tag is severely limited. In contrast to the jellyfish fluorescent proteins, which have been successfully used to tag hundreds of proteins, DsRed conjugates have proven much less successful and are often toxic.
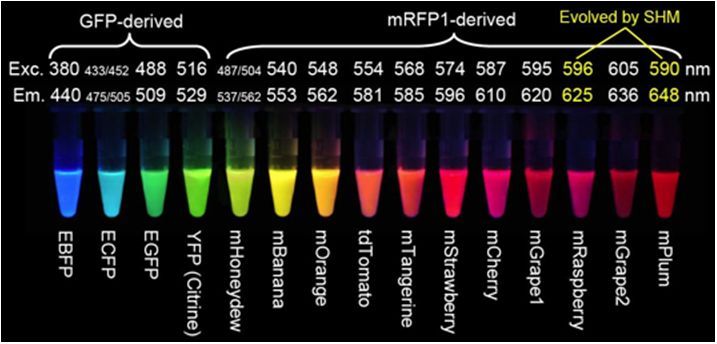
A few of the problems with DsRed fluorescent proteins have been overcome through mutagenesis. The second-generation DsRed, known as DsRed2, contains several mutations at the peptide amino terminus that prevent formation of protein aggregates and reduce toxicity. In addition, the fluorophore maturation time is reduced with these modifications. The DsRed2 protein still forms a tetramer, but it is more compatible with green fluorescent proteins in multiple labeling experiments due to the quicker maturation. Further reductions in maturation time have been realized with the third generation of DsRed mutants, which also display an increased brightness level in terms of peak cellular fluorescence. Red fluorescence emission from DsRed-Express can be observed within an hour after expression, as compared to approximately six hours for DsRed2 and 11 hours for DsRed. A yeast-optimized variant, termed RedStar, has been developed that also has an improved maturation rate and increased brightness. The presence of a green state in DsRed-Express and RedStar is not apparent, rendering these fluorescent proteins the best choice in the orange-red spectral region for multiple labeling experiments. Because these probes remain obligate tetramers, they are not the best choice for labeling proteins.
红色荧光蛋白的激发波长和发射波长Red Proteins
| Protein | Excitation Wavelength | Emission Wavelength |
|---|---|---|
| TagRFP | 555 | 584 |
| TagRFP-T | 555 | 584 |
| RRvT | 556 | 583 |
| mRuby | 558 | 605 |
| mRuby2 | 559 | 600 |
| mTangerine | 568 | 585 |
| mApple | 568 | 592 |
| mStrawberry | 574 | 596 |
| FusionRed | 580 | 608 |
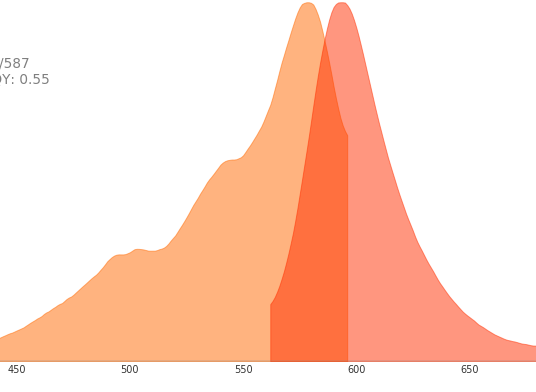
| mCherry | 587 | 610 |
| mNectarine | 558 | 578 |
| mRuby3 | 558 | 592 |
| mScarlet | 569 | 594 |
| mScarlet-I | 569 | 593 |